Reversing the Rust: How N-Acetylcysteine (NAC) Repairs and Protects Your Cells
A Master Antioxidant Precursor
How N-Acetylcysteine (NAC) Repairs and Protects Your Cells
Abbreviations
GSH: Glutathione (Reduced form, active antioxidant)
IL-6: Interleukin-6 (Inflammatory cytokine)
NAC: N-Acetylcysteine
NF-κB: Nuclear Factor kappa B (Master inflammation pathway)
ROS: Reactive Oxygen Species
TNF-α: Tumor Necrosis Factor alpha (Inflammatory signaling molecule)
1.N-acetylcysteine NAC Use as an Antioxidant
Mechanisms of Action
N-acetylcysteine (NAC) is a widely utilized, highly versatile synthetic antioxidant and an acetylated derivative of the naturally occurring amino acid L-cysteine. Characterized by a molecular weight of 163.2 g/mol and a characteristically sour taste, NAC has been rigorously studied and recognized for decades as an essential medicine by the World Health Organization (WHO), largely due to its remarkable safety profile and low cost. Originally synthesized, patented, and deployed as an inhaled mucolytic agent designed to manage and break down heavily cross-linked mucus in chronic respiratory conditions like chronic obstructive pulmonary disease (COPD) and cystic fibrosis, NAC’s primary biochemical utility extends far beyond pulmonology, lying in its robust systemic anti-inflammatory and free-radical scavenging capabilities.
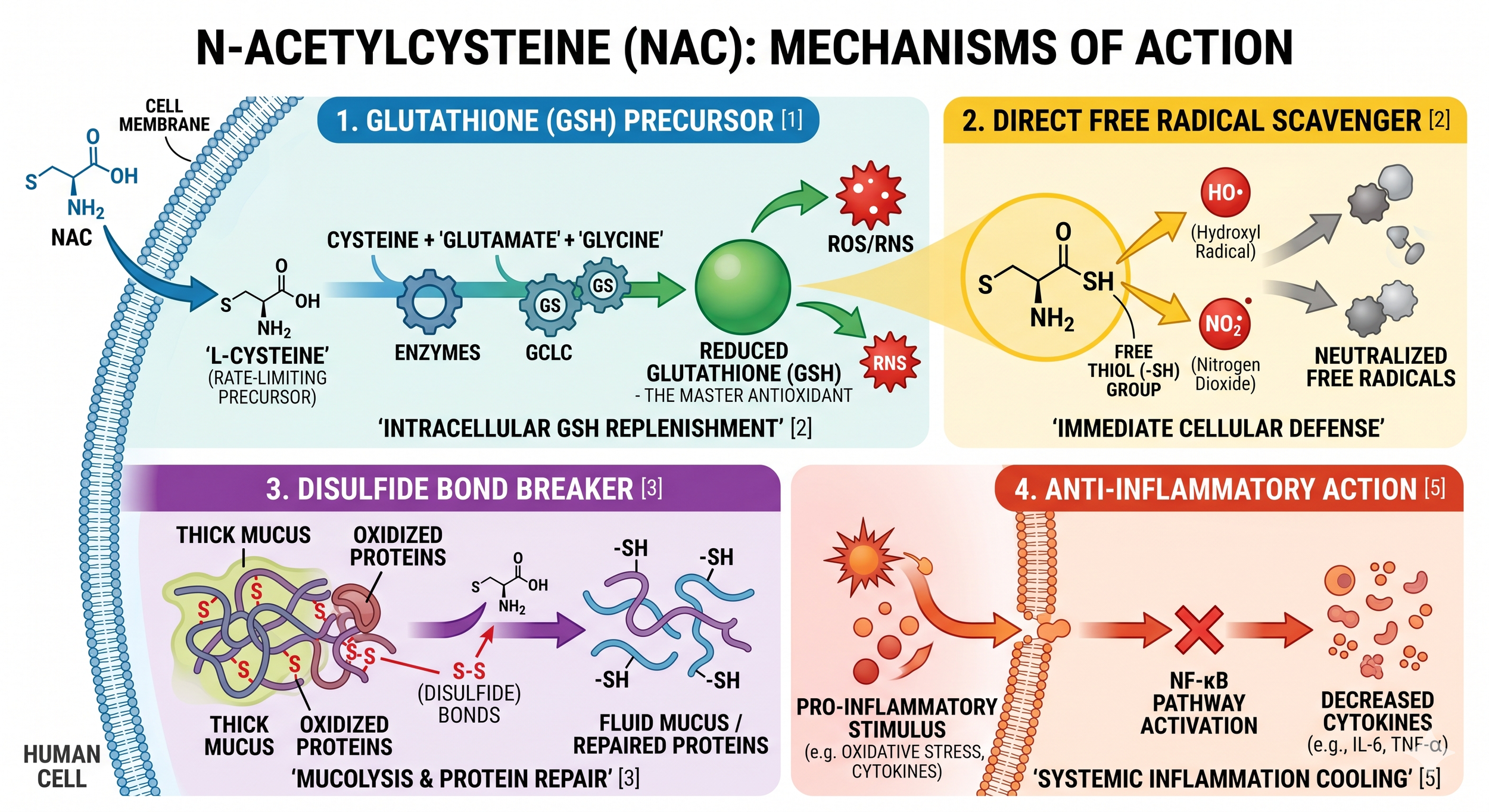
The compound operates fundamentally through two distinct yet complementary biochemical pathways: indirect, glutathione-dependent mechanisms, and direct, glutathione-independent mechanisms. Indirectly, NAC acts as a highly efficient, membrane-permeable prodrug for L-cysteine. Intracellularly, it undergoes rapid deacetylation to replenish cysteine, which is the crucial, rate-limiting substrate required for the biosynthesis of glutathione (GSH)—the human body's most critical endogenous antioxidant biothiol (1). By dramatically elevating intracellular GSH levels, NAC supports the enzymatic neutralization of reactive oxygen species via powerful enzymes such as glutathione peroxidase (GPx) and glutathione-S-transferase (GST) (2). This indirect GSH-replenishment pathway represents the foundational biochemical rationale for its use as the absolute gold-standard antidote for acetaminophen (paracetamol) poisoning. In cases of intentional or accidental overdose, the hepatic GSH pool is rapidly and entirely depleted by the highly toxic acetaminophen metabolite N-acetyl-p-benzoquinone imine (NAPQI). Massive intravenous or oral doses of NAC are administered to rapidly restore hepatic GSH, which then binds to and neutralizes the NAPQI toxin before irreversible, fatal hepatic necrosis can occur. (3)
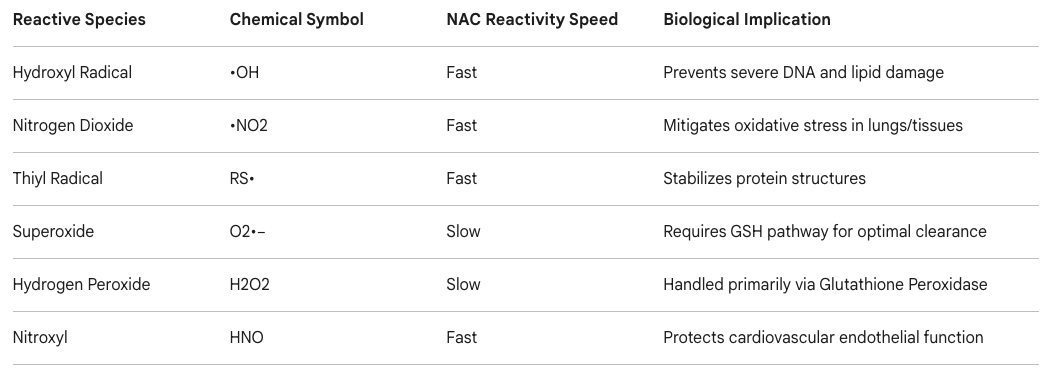
Directly, the unique molecular structure of NAC features a nucleophilic sulfhydryl (-SH) group. This specific functional group allows NAC to act as an independent scavenger of certain electrophilic free radicals and oxidant species without needing to be converted into GSH (1). Under controlled experimental parameters, the free thiol group of NAC reacts directly and rapidly with the hydroxyl radical (•OH), nitrogen dioxide (•NO2), the carbon trioxide ion (CO3•−), and the thiyl radical (RS•), while reacting somewhat more slowly with superoxide (O2•−) and hydrogen peroxide (H2O2) (2). This direct scavenging effectively mitigates lipid peroxidation and is highly instrumental in preventing cellular ferroptosis during acute tissue injury (3).
Table 1: Reactivity profile of the un-metabolized N-acetylcysteine sulfhydryl group against various reactive oxygen and nitrogen species (1).
2.Current Clinical Uses
Furthermore, NAC possesses profound disulfide-breaking activity. It chemically cleaves the robust disulfide bridges within high-molecular-weight mucus glycoproteins, dramatically reducing their viscosity and allowing for pulmonary clearance (1). Beyond the respiratory and hepatic systems, NAC demonstrates profound modulatory effects on the central nervous system. It easily permeates the blood-brain barrier—where standard oral GSH administration completely fails due to rapid gastrointestinal and hepatic hydrolysis—upregulating the Nrf2-ARE antioxidant transcription pathway (2). It is also critical in restoring glutamate homeostasis by enhancing the activity of the GLT-1 transporter on astrocytes, which clears toxic excess glutamate from the synaptic cleft, and by stimulating the expression of Brain-Derived Neurotrophic Factor (BDNF) to promote neuronal survival, neurogenesis, and synaptic plasticity (2).
Essential Findings
Glutathione Precursor: NAC's principal systemic mechanism of action involves functioning as a cell-permeable prodrug for L-cysteine. Once inside the cytoplasm, it drives the rapid biosynthesis and replenishment of glutathione (GSH), the body's master antioxidant, which is frequently and severely depleted during chronic disease states, natural aging, and xenobiotic intoxication.
Direct Free-Radical Scavenging: Completely independent of GSH synthesis, NAC's free thiol (-SH) group acts as a potent chemical nucleophile. It reacts directly and rapidly with specific, highly toxic free radicals—such as nitrogen dioxide, hydroxyl radicals, and lipid peroxides—providing immediate cytoprotection against acute oxidative tissue damage.
Disulfide Breaking and Mucolytic Action: The sulfhydryl group in NAC breaks the complex disulfide bonds in heavy mucus glycoproteins (mucins). This reduces the physical viscosity of tenacious mucous secretions, forming the established biochemical basis for its efficacy in treating COPD, cystic fibrosis, and other severe obstructive airway diseases.
Acetaminophen Antidote Mechanism: In paracetamol-induced hepatotoxicity, NAC prevents acute liver failure by replenishing the hepatic glutathione pool. This newly synthesized GSH reacts directly with NAPQI, the toxic, electrophilic metabolite of acetaminophen, effectively neutralizing it before it can destroy the structural hepatocyte (1).
Nrf2-ARE Pathway Activation: NAC actively induces the Nrf2 (Nuclear factor erythroid 2-related factor 2) cellular signaling pathway. This critical activation upregulates the nuclear transcription of endogenous antioxidant enzymes, including glutathione reductase, glutathione peroxidase, and heme oxygenase-1 (HO-1), vastly expanding the cell's internal defense network against oxidative stress (1).
Neurotransmitter Regulation: Within the central nervous system, NAC restores vital glutamate homeostasis. It accomplishes this by enhancing the activity of the GLT-1 transporter on astrocytes, which rapidly clears excitotoxic excess glutamate from the synaptic cleft, and by interacting with type II metabotropic glutamate receptors (mGluR2/3). This mechanism has vast clinical implications for psychiatric conditions linked to neurotransmitter dysregulation, including schizophrenia and severe addiction (1).
Neurotrophic and Barrier Support: NAC administration leads to the therapeutic elevation of Brain-Derived Neurotrophic Factor (BDNF) via the TrkB receptor signaling pathway, enhancing overall neurogenesis. Additionally, it stabilizes the blood-brain barrier by modulating astrocytic gap junctions (connexin-37 and 43) and closely managing the localization of tight junction proteins like claudin-5 (1).
Sulfane Sulfur Metabolism: Emerging biochemical evidence indicates that a portion of NAC's GSH-independent antioxidant capacity is derived from its rapid cellular metabolism into highly reactive sulfane sulfur compounds, which provide unique, rapid-acting neutralization of reactive oxygen species prior to the completion of the GSH synthesis cycle (1).
Figure 1. The Four Core Mechanisms of N-Acetylcysteine (NAC) in Cellular Defense and Repair.
3.Clinical Summary
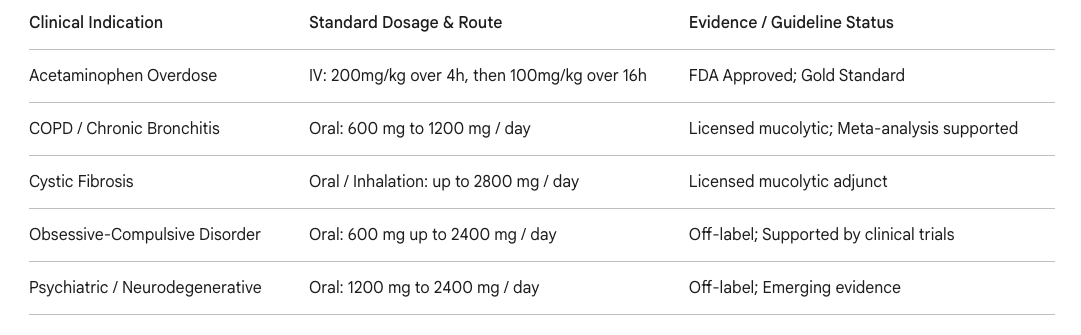
In routine and specialized clinical practice, N-acetylcysteine possesses a uniquely broad therapeutic index and highly diverse applications owing to its dual functionality as a direct chemical reducing agent and an indirect metabolic amino acid precursor. Its most established, FDA-approved clinical role is as the definitive, life-saving antidote for acute acetaminophen toxicity, where timely intravenous or oral administration—optimally initiated within 8 hours of drug ingestion—exhibits a near 100% efficacy rate in preventing acute, irreversible hepatic failure. As a mucolytic agent, international guidelines heavily support its long-term use in respiratory medicine. Comprehensive meta-analyses demonstrate that daily oral doses of 600 mg significantly benefit chronic bronchitis patients without major airway obstruction, whereas patients presenting with documented, severe airway obstruction require higher dosages (≥1200 mg daily) to effectively prevent acute respiratory exacerbations.
Beyond its primary, traditional indications, NAC's exceptional safety profile, high tolerability, and low economic cost have spurred massive global investigations into its use as an adjunctive therapy for a wide array of chronic inflammatory, psychiatric, and metabolic conditions (1). The compound's systemic protein binding is relatively high (typically 66–97% bound to albumin) (2). It exhibits substantial clinical potential in managing systemic conditions intimately associated with profound oxidative stress, such as polycystic ovary syndrome (PCOS), male infertility, obsessive-compulsive disorder (OCD), bipolar depression, and progressive neurodegenerative diseases (3).
Table 2: Summary of common FDA-approved and off-label clinical indications for N-acetylcysteine, alongside standard dosing parameters (1).
4.Lay Summary
N-acetylcysteine, commonly referred to as NAC, is a synthetic, supplement form of a natural amino acid that acts as an incredibly versatile medicinal tool in the human body. It possesses two main biochemical superpowers. First, it acts as an express delivery vehicle for the raw materials your body desperately needs to build a molecule called "glutathione." Glutathione is often referred to by scientists as the body’s "master antioxidant" because it protects the liver, brain, and other vital organs from being permanently damaged by toxic chemicals and everyday cellular wear and tear. If someone accidentally or intentionally overdoses on Tylenol (acetaminophen), their liver completely runs out of glutathione and rapidly begins to shut down. To reverse this, emergency room doctors use massive intravenous doses of NAC to rapidly rebuild the liver's glutathione defenses, neutralizing the Tylenol toxins and saving the patient's life.
Second, NAC has a highly unique chemical shape that allows it to directly attack certain toxins completely on its own, without waiting to be turned into glutathione. More importantly, this shape gives it the ability to chemically "cut" through thick, sticky protein substances. This is exactly why it has been used for decades as a prescription medication for patients with severe lung diseases like chronic bronchitis and cystic fibrosis—NAC acts like a pair of chemical scissors, chopping up thick, suffocating mucus in the lungs so it can be easily coughed up by the patient. Because it is highly safe, inexpensive, and exceptionally good at fighting inflammation and protecting cells, medical researchers are now successfully utilizing NAC to treat a wide variety of other difficult health problems, ranging from mental health disorders like obsessive-compulsive disorder (OCD) and addiction, to complex chronic inflammatory and metabolic diseases.
Reference List
Tenório Mda C, et al. N-Acetylcysteine: A Review of Clinical Usefulness. Journal of Nutrition and Metabolism. 2021.
Chen CH, et al. N-Acetylcysteine Alleviates the Progression of Chronic Kidney Disease: A Three-Year Cohort Study. Medicina. 2023;59(11).
Aparicio-Trejo OE, et al. NAC administration prevented mitochondrial alterations in both organs and heart damage. Antioxidants. 2024;13.
Ndumele CE, et al. A Synopsis of the Evidence for CKM Syndrome. Circulation. 2023.
Sabetghadam M, et al. Therapeutic effects of oral NAC in patients with acute ischemic stroke: A randomized trial. Therapeutic Advances in Neurological Disorders. 2020.