Therapeutic Efficacy of N-Acetylcysteine in Cardiovascular-Kidney-Metabolic (CKM) Syndrome and Ischemic Stroke
A Master Antioxidant Precursor - Part 2
How N-Acetylcysteine (NAC) Repairs and Protects Your Cells
Abbreviations
ACE / ARB: Angiotensin-Converting Enzyme / Angiotensin Receptor Blocker
AHA: American Heart Association
BMI: Body Mass Index
CKD: Chronic Kidney Disease
CKM: Cardiovascular-Kidney-Metabolic
CRS-IV: Cardio-Renal Syndrome Type 4
CVD: Cardiovascular Disease
eGFR: Estimated Glomerular Filtration Rate
ESRD: End-Stage Renal Disease
GCS: Glasgow Coma Scale
GLP-1 RA: Glucagon-Like Peptide-1 Receptor Agonist
ICU: Intensive Care Unit
IL-6: Interleukin-6
mRS: Modified Rankin Scale
NIHSS: National Institutes of Health Stroke Scale
NSE: Neuron-Specific Enolase
S100B: S100 calcium-binding protein B
SCr: Serum Creatinine
SGLT2i: Sodium-Glucose Cotransporter-2 Inhibitor
SIRT1/3: Sirtuin 1 and 3 (Enzymes that regulate longevity and energy)
1.N-acetylcysteineNAC Use as an Antioxidant
Mechanisms of Action
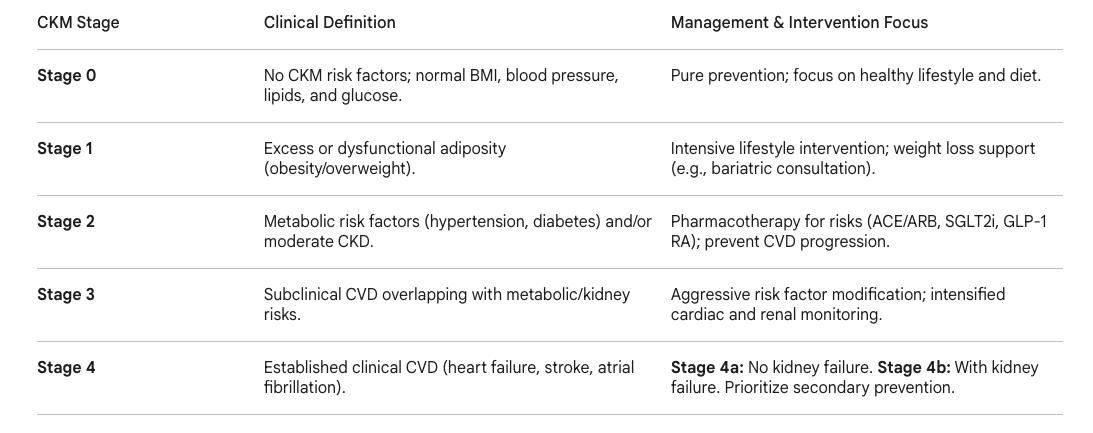
Cardiovascular-Kidney-Metabolic (CKM) syndrome represents a newly formalized, highly integrated, and urgent pathophysiological framework officially recognized by the American Heart Association (AHA) in a 2023 Presidential Advisory (1,2).This systemic disorder fundamentally acknowledges the reciprocal, deeply deleterious connections among excess or dysfunctional adiposity, Type 2 diabetes, chronic kidney disease (CKD), and clinical cardiovascular disease (CVD) (2).The AHA classifies CKM syndrome across a progressive, five-stage model designed to capture the continuum of the disease and target interventions accordingly.
Table 1: The American Heart Association (AHA) Cardiovascular-Kidney-Metabolic (CKM) Syndrome Staging Model and associated clinical focuses(1,2).
2.Current Clinical Uses
A central biological driver propelling the catastrophic multi-organ dysfunction inherent in CKM progression is profound, systemic oxidative stress. This oxidative burden initiates mitochondrial bioenergetic failure, chronic low-grade inflammation, and severe endothelial dysfunction (1). Specifically, early-stage CKM pathophysiology is marked by the onset of Cardio-Renal Syndrome Type 4 (CRS-IV). In CRS-IV, primary renal mitochondrial impairment cascades to induce systemic inflammation via the hyperactivation of the NLRP3 inflammasome and NF-κB pathways. This triggers secondary cardiac mitochondrial damage, the uncoupling of oxidative phosphorylation, and subsequent cardiac hypertrophy and heart failure (2). Consequently, potent antioxidant interventions, particularly N-acetylcysteine (NAC), have emerged as highly promising, multi-organ therapeutic agents designed to halt this systemic deterioration at the cellular level.
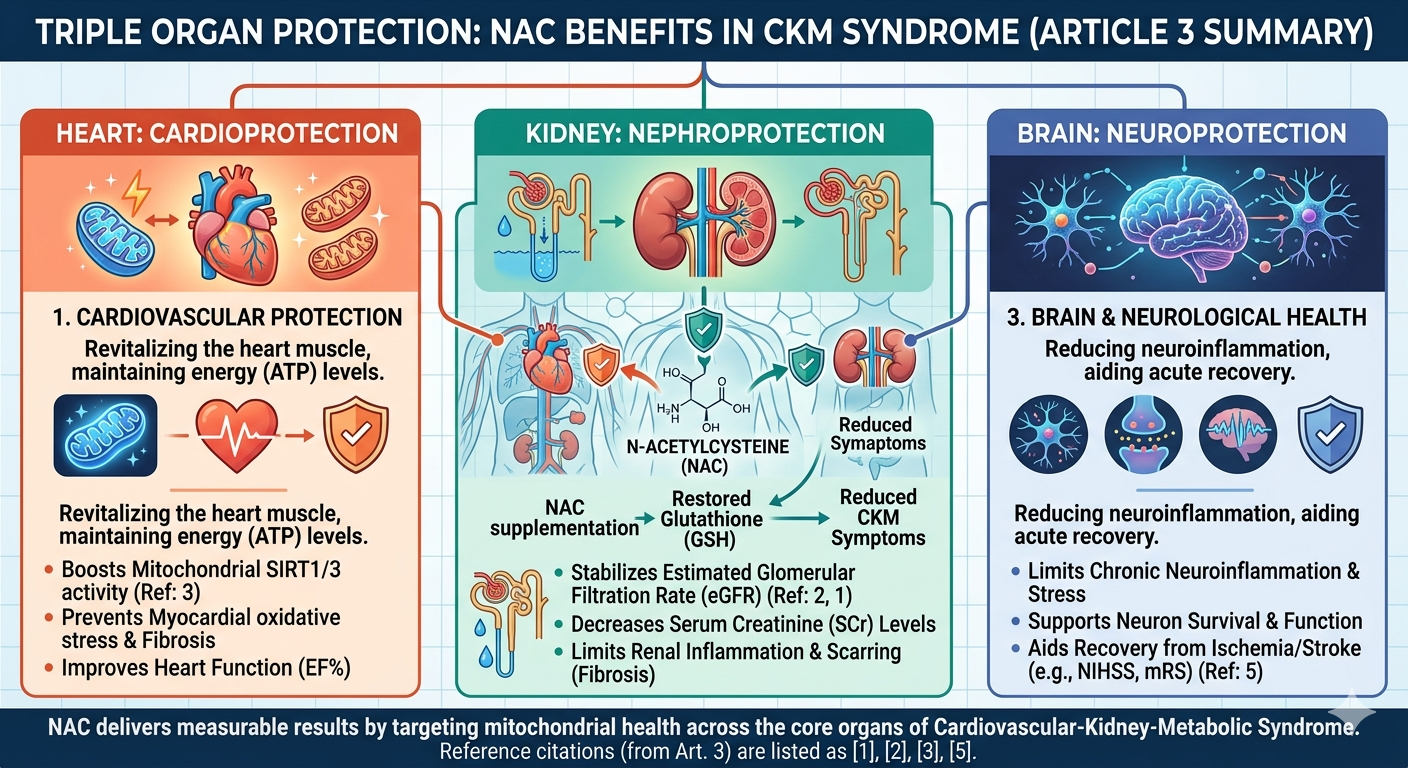
Emerging Multisystem Shield
In the renal/ kidney system, NAC provides comprehensive protection by upregulating crucial mitochondrial biogenesis through key intracellular second messenger systems, the AMPK-SIRT1/3-PGC-1α signaling pathway. By binding to and activating sirtuins (specifically SIRT1 and SIRT3), NAC preserves the mitochondrial membrane potential and complex respiratory activity (3). This drastically reduces the release of uremic toxins and inflammatory cytokines (like IL-1β and IL-6) into the systemic circulation, severing the toxic crosstalk between the kidneys and the heart (1). This specific mechanism halts the progression of CRS-IV and directly protects the myocardium from secondary failure (1).
In the central nervous system, which often suffers catastrophic outcomes during Stage 4 CKM in the form of acute ischemic stroke, NAC provides dual-action cerebral protection. Experimental and human clinical data suggest that NAC acts not only as a profound neuroprotectant—scavenging ROS, preventing ferroptosis, and drastically reducing post-ischemic neuroinflammation—but also possesses intrinsic, potent thrombolytic properties. NAC is uniquely capable of cleaving von Willebrand factor multimers, thereby dissolving both mixed and platelet-rich thrombi to facilitate rapid arterial recanalization (5). Through these combined renal, cardiac, and cerebral mechanisms, NAC positions itself as a critical, multi-system shield capable of disrupting the complex biological feedback loops that drive mortality in advanced CKM syndrome. The AHA defines CKM syndrome as a systemic disorder driven by connections among obesity, diabetes, CKD, and cardiovascular disease, significantly increasing the risk of premature all-cause and cardiovascular mortality, especially in stages 3 and 4 (1).
Essential Findings
Cardio-Renal Syndrome Protection (CRS-IV): Advanced animal models of CRS-IV (induced via 5/6 nephrectomy) conclusively demonstrate that pre-administration of NAC prevents the early onset of fatal mitochondrial alterations in both the heart and kidneys (3). It operates by hyper-activating the SIRT1/3-PGC-1α pathway, inducing rapid mitochondrial biogenesis, and restoring the redox state, which actively prevents downstream cardiac hypertrophy and ejection fraction decline (1).
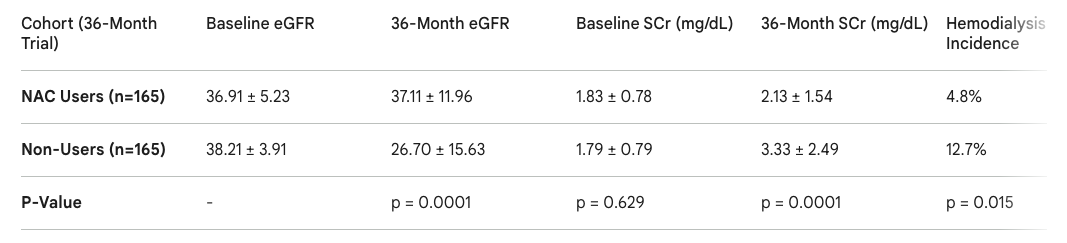
Clinical Efficacy in Chronic Kidney Disease: In a highly detailed, 36-month retrospective cohort study utilizing the National Health Insurance database in Taiwan, tracking 7668 CKD patients, prolonged NAC administration significantly arrested renal decline (4). Matched cohorts revealed that patients consuming NAC for three years maintained nearly baseline levels of estimated Glomerular Filtration Rate (eGFR) and Serum Creatinine (SCr), whereas non-users experienced severe, statistically significant functional deterioration.
Prevention of Hemodialysis in CKD: Long-term NAC utilization provides profound, life-altering clinical endpoints in CKD populations. In the 36-month study, the incidence rate of progression to End-Stage Renal Disease (ESRD) requiring hemodialysis was 2.6 times lower in the NAC cohort (4.8%) compared directly to the unmedicated control group (12.7%), presenting a striking odds ratio of 34.9 (higher odds of an outcome)(4).
DKD vs. Non-DKD Variability: The therapeutic efficacy of NAC in CKD shows strong etiology-dependent variation. While NAC effectively stabilizes eGFR and SCr in patients suffering from non-diabetic kidney disease (non-DKD), patients with Diabetic Kidney Disease (DKD) still experience progressive renal decline despite continuous NAC treatment. This indicates that the severe, compounding metabolic derangements inherent to diabetes may overpower NAC's specific antioxidant capacity (1).
Cerebral Protection and Stroke Recovery: In cases of acute ischemic stroke (a hallmark of Stage 4 CKM), early NAC administration demonstrates highly tangible clinical benefits. Randomized controlled human trials indicate that both high-dose intravenous NAC (standardized to 200mg/kg loading dose over 4 hours, followed by 100mg/kg over 16 hours) and intensive oral dosing regimens significantly reduce long-term neurological deficits (7). Patients receiving NAC showed vastly superior improvements in both National Institutes of Health Stroke Scale (NIHSS) and modified Rankin Scale (mRS) scores at 90 days post-infarction (6).
Reduction of Neurological Biomarkers: NAC treatment in acute stroke patients drastically suppresses the spike in acute neuroinflammation and brain injury biomarkers. Trial data highlights significant, precipitous declines in circulating Interleukin-6 (IL-6), Neuron-Specific Enolase (NSE), and S100 calcium-binding protein B (S100B) by day 7 post-infarction, which correlates directly with improved Glasgow Coma Scale (GCS) outcomes and shorter ICU stays (6,7).
Intergenerational CKM Programming: Maternal exposure to high-fat diets frequently predisposes offspring to severe CKM phenotypes (including hypertension, kidney disease, and dyslipidemia) later in life. Strikingly, providing continuous NAC (500 mg/kg/day) to mothers during gestation and lactation completely prevents the developmental programming of hypertension and kidney disease in the offspring by resolving the underlying maternal oxidative stress (8).
Figure 1. The Triple Organ Protective Effects of N-Acetylcysteine (NAC) in Cellular Defense and Repair.
3.Clinical Summary
The integration of N-acetylcysteine into the clinical management algorithms of Cardiovascular-Kidney-Metabolic syndrome represents a necessary paradigm shift toward treating the root oxidative pathology explicitly linking these organ systems. Clinicians must view CKM not as isolated, parallel conditions, but as a deeply integrated biological continuum where kidney dysfunction dramatically and predictably amplifies cardiovascular and metabolic risk (1).For patients identified in Stage 2 and Stage 3 CKM (characterized by moderate CKD and subclinical CVD), the administration of oral NAC can serve as a potent, highly affordable adjunct to standard, guideline-directed medical therapies such as ACE inhibitors, ARBs, and SGLT2 inhibitors.
Long-term outcome data is exceptionally compelling for renal preservation; prescribing oral NAC over a multi-year horizon (≥ 36 months) significantly preserves the eGFR and stabilizes serum creatinine (4).
Table 1: Comparative 36-month renal outcomes between NAC users and matched non-users in a Stage 3-5 CKD cohort, highlighting significant preservation of renal function and avoidance of ESRD in the NAC group (4).
This protocol markedly reduces the absolute risk of progression to hemodialysis, particularly in non-diabetic populations where the primary driver of decline is oxidative and hemodynamic rather than advanced glycation (4).
In the acute intensive care setting of Stage 4 CKM—specifically during an acute ischemic stroke—high-dose intravenous NAC has successfully translated from experimental neuroprotection models directly to human efficacy. Intravenous protocols mirroring the established acetaminophen toxicity dosing (200 mg/kg over 4 hours, followed by 100 mg/kg over 16 hours) have proven exceptionally safe and highly effective at limiting infarct expansion (7).It mitigates secondary neuroinflammation (evidenced by crashing IL-6 and NSE levels) and vastly improves 90-day functional independence metrics (6).
Importantly, the safety profile of NAC across these high-risk, fragile CKM populations remains highly favorable. In patients with severe chronic kidney disease, NAC does not require complex dosage adjustments and acts as a safe intervention devoid of significant nephrotoxic or hepatotoxic side effects (9). Standard oral administration (600–1200 mg daily) is well tolerated, with adverse events largely restricted to mild, transient gastrointestinal complaints such as nausea or dyspepsia (9). When utilizing rapid intravenous NAC protocols in the ICU for cerebral or cardiac protection, clinicians must continuously monitor for rare anaphylactoid reactions—such as acute flushing, pruritus, or potentially hazardous bronchoconstriction—while noting that true, IgE-mediated allergic anaphylaxis remains statistically anomalous and is generally rate-dependent (10).
4.Lay Summary
Medical experts and cardiologists recently grouped heart disease, kidney disease, diabetes, and obesity into a single, highly interconnected web of illness called "Cardiovascular-Kidney-Metabolic (CKM) Syndrome." They realized that treating these diseases separately in isolation simply doesn't work well because a failure in one organ quickly causes a catastrophic crash in the others. For example, when the body's filters (the kidneys) become damaged by high blood pressure or diabetes, they release highly inflammatory toxins into the blood. These toxins travel directly to the heart muscle, causing it to swell and weaken—a chain reaction driven by massive amounts of cellular oxidative stress.
N-acetylcysteine (NAC) acts as a powerful, systemic shield that can successfully interrupt this deadly chain reaction. By protecting the microscopic engines (called mitochondria) inside both kidney and heart cells, NAC literally stops the kidneys from releasing heart-damaging toxins. Long-term medical studies analyzing thousands of patients show that individuals with kidney disease who take NAC regularly for several years can keep their kidney function remarkably stable. Most importantly, taking NAC drastically lowers their chances of their kidneys failing completely and ever needing to be hooked up to a dialysis machine.
Furthermore, if a patient reaches the most severe, dangerous stage of CKM syndrome and suffers a stroke (a blockage of blood to the brain), NAC can be used as emergency medicine to protect the brain tissue from dying. When given through an IV drip shortly after a stroke occurs, NAC acts to rapidly cool down severe, destructive inflammation in the brain and can even help chemically dissolve the blood clots causing the blockage. Patients who receive this specific IV treatment show far less brain damage on their hospital blood tests and recover their motor functions, speech, and daily independence much faster than those who do not. Ultimately, because NAC is incredibly safe, highly affordable, and targets the exact root oxidative damage connecting all these vital organs, it is rapidly becoming a vital, multi-purpose tool for protecting the heart, kidneys, and brain against the ravages of aging and disease.
Reference List
Ndumele, C.E. et al. "A synopsis of the evidence for the science and clinical management of Cardiovascular-Kidney-Metabolic (CKM) Syndrome." Circulation, 2023.
American Heart Association. "Top 10 Highlights: 2023 Cardiovascular-Kidney-Metabolic Health Presidential Advisory." AHA Professional, 2023.
Tapia, E. et al. "Mitochondrial Bioenergetics, Biogenesis, and Redox Impairment in Cardio-Renal Syndrome Type 4: Protective Role of N-Acetylcysteine." Antioxidants, 2025.
Hsu, Y-J. et al. "N-Acetylcysteine Alleviates the Progression of Chronic Kidney Disease." Medicina, 2023.
Martinez de Lizarrondo, S. et al. "Potent Thrombolytic Effect of N-Acetylcysteine on Arterial Thrombi." Circulation, 2017.
Fallahzadeh, M.A. et al. "Effect of oral N-acetylcysteine on neurological outcomes in patients with acute ischemic stroke." Journal of Research in Medical Sciences, 2020.
Ali, A. et al. "Clinical Trials of Intravenous N-Acetylcysteine for the Treatment of Acute Ischemic Stroke." ClinicalTrials.gov, 2021.
Tain, Y-L. et al. "Maternal High-Fat Diet and Oxidative Stress in Cardiovascular-Kidney-Metabolic Syndrome Programming." Nutrients, 2024.
Tenório Mda C, et al. "N-Acetylcysteine (NAC): Impacts on Human Health." Antioxidants, 2021. (Note: This is the definitive modern review covering the general safety, lack of major organ toxicity, and mild GI side effects of oral NAC).
Sandilands, E.A. and Bateman, D.N. "Adverse reactions associated with acetylcysteine." Clinical Toxicology, 2009.