The Pathophysiology of Oxidative Stress and Age-Related Disease
Mitochondrial Dysfunction in Aging and Disease -
The “Rust-Effect”on Essential Organs
Abbreviations
AUC: Area Under the Curve
CAT: Catalase
CKD: Chronic Kidney Disease
DAMPs: Damage-Associated Molecular Patterns
LIP: Labile Iron Pool
MDA: Malondialdehyde
MPO: Myeloperoxidase
NADPH: Nicotinamide Adenine Dinucleotide Phosphate
NAFLD: Non-Alcoholic Fatty Liver Disease
NASH: Non-Alcoholic Steatohepatitis
NF-κB: Nuclear Factor kappa B
RNS: Reactive Nitrogen Species
ROC: Receiver Operating Characteristic
ROS: Reactive Oxygen Species
VDACs: Voltage-Dependent Anion Channels
1.Aging - Resisting Acceleration
Excessive Oxidation
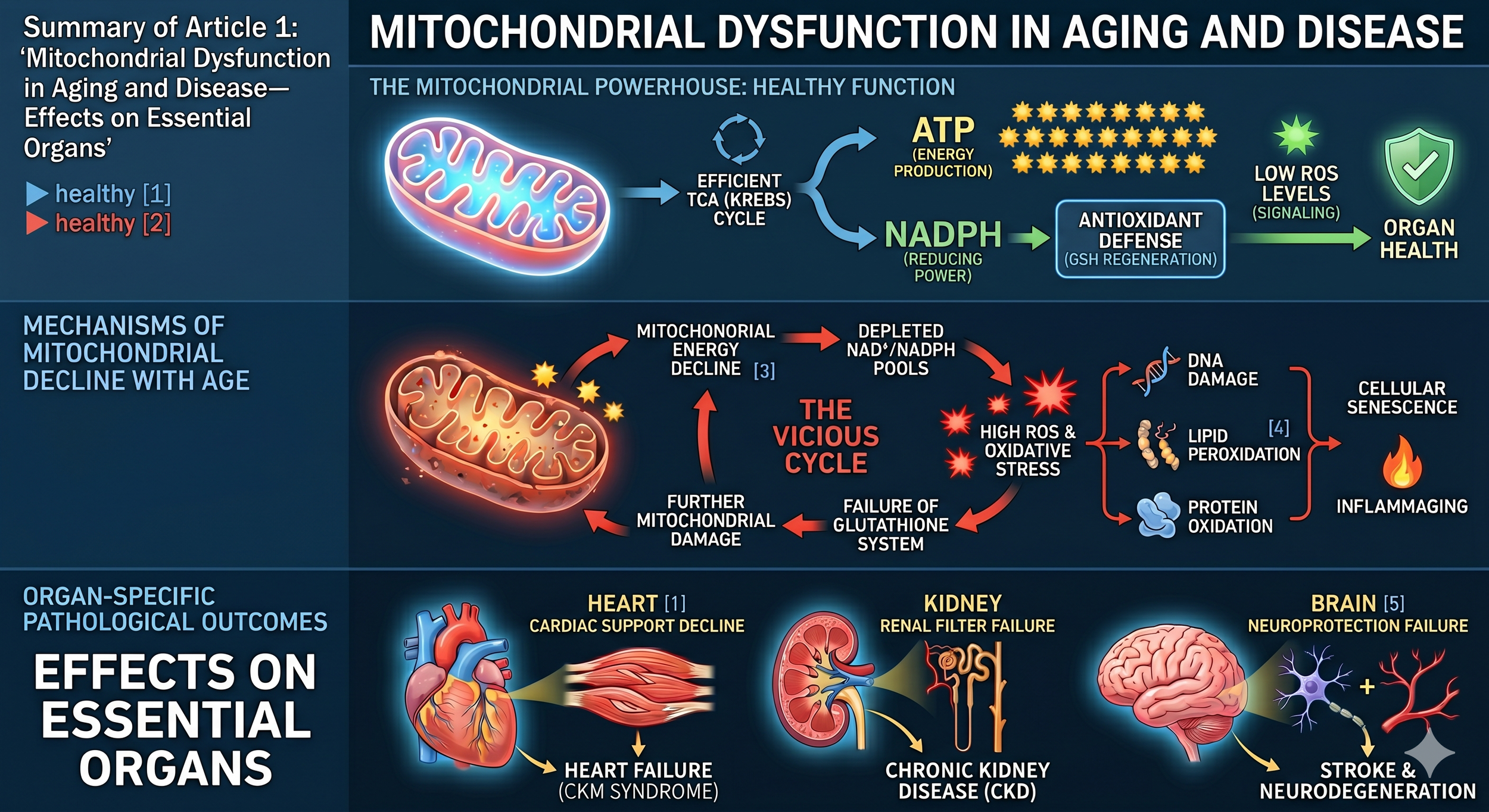
Aging is fundamentally characterised as a normal, inevitable, irreversible, and progressive biological decline driven by a highly complex interplay of internal physiological processes and external environmental factors(1). At the core of cellular senescence, tissue degradation, and the induction of age-related morbidity is the phenomenon of oxidative stress. Oxidative stress is defined as a critical and deleterious imbalance between the generation of highly reactive oxidant species—namely, reactive oxygen species (ROS) and reactive nitrogen species (RNS)—and the intrinsic cellular antioxidant defense machinery designed to neutralize the(2). Under normal physiological conditions, low concentrations of ROS are not merely toxic byproducts but serve as essential signaling molecules that facilitate crucial adaptations, such as skeletal muscle conditioning post-exercise, cellular proliferation, and immune responses (3). However, when internal physiological triggers combine with external environmental stressors, the overproduction of free radicals catastrophically overwhelms the body's redox homeostasis (the balance between oxidative and reductive reactions within cells essential for many chemical interactions) (1).
The intrinsic triggers of oxidative stress primarily originate within the cellular mitochondria. The mitochondrial electron transport chain (specifically Complexes I and III) - [This is the power plant and part of the energy production cycle in all animal living cells. ] - frequently leaks electrons during the production of adenosine triphosphate (ATP), leading to the premature reduction of oxygen and the formation of the superoxide anion. This process is exacerbated by increased nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity, which actively generates ROS as part of the immune response (2). When a healthy body is invaded by a bacteria or virus, immune cells (like macrophages) rush to the site. They activate the NADPH oxidase enzyme to deliberately generate a massive burst of Reactive Oxygen Species (ROS), like a flame thrower. In this context, ROS is used as a highly toxic chemical weapon to destroy the invading pathogens. Once the infection is cleared, the enzyme shuts off, and the body's antioxidants (powered by regular NADPH) clean up the collateral damage.
The Problem with Aging and Disease: The "Double Whammy"
As we age, or when we develop metabolic diseases (like in CKM syndrome), the body enters a state of chronic, low-grade inflammation (often called "inflammaging").
Because of this chronic inflammation, the immune system thinks it is constantly under attack. As a result, the NADPH oxidase enzymes get stuck in the "ON" position. This causes a catastrophic "double whammy" for the cell:
Massive Damage: The overactive enzymes constantly pump out high levels of ROS, destroying healthy tissues, mutating DNA, and damaging the kidneys, heart, and brain.
Fuel Depletion: Because NADPH oxidase consumes NADPH to create this damage, it rapidly drains the cell's limited pool of NADPH. It steals the very "repair fluid" that the mitochondria desperately need to regenerate glutathione.Concurrently, external triggers such as airborne pollutants, ultraviolet (UV) radiation, tobacco smoke, heavy metals, and hypercaloric diets introduce exogenous free radicals or stimulate their endogenous production (1). This convergence of factors results in severe biomolecular damage, precipitating the oxidation of cellular lipids, structural proteins, and nucleic acids (DNA/RNA)(3). The oxidation of lipid membranes disrupts cellular integrity, while DNA oxidation leads to mutagenic alterations, serving as the primary etiology for a broad spectrum of chronic diseases and the acceleration of the biological aging process (3).
Figure 1: Schematic representation of mitochondrial dysfunction and oxidative stress that leads to damage to essential organs as we age and accelerate in some disease states such as chromic kidney disease .
2.Vicious Circles
A critical mechanism in this pathological cascade is the establishment of a vicious, self-sustaining feedback loop between chronic oxidative stress and systemic inflammation. The immune system is activated by the presence of oxidatively modified proteins and lipids, recognizing them as damage-associated molecular patterns (DAMPs). This recognition induces an inflammatory state characterized by the release of cytokines and the activation of macrophages, which in turn generate further ROS as a defensive mechanism, thereby significantly increasing age-related morbidity and mortality (4). The systemic nature of oxidative stress dictates that its pathogenesis manifests uniquely across different organ systems, leading to highly diverse clinical presentations. In the hematologic (blood) system, for example, oxidative stress compromises the structural integrity of erythrocytes (red blood cells). The oxidation of red blood cell lipid membranes decreases their elasticity, shortens their physiological lifespan, and drastically increases the probability of hemolysis. This mechanism explains the high prevalence and onset of anemia in patients with chronic kidney disease (CKD), compounding the secondary effects of reduced erythropoietin synthesis (4).
Can You Have Too Much Iron ?
The deregulation of cellular iron homeostasis plays a pivotal role in oxidative damage. Iron is essential for biological systems due to its ability to rapidly switch between Fe2+ and Fe3+ states; however, when unbound to proteins, this same chemical property makes it paradoxically dangerous (5). Dysregulation expands the labile iron pool (LIP), catalyzing the formation of highly reactive hydroxyl radicals through Fenton chemistry (5). This specific pathway initiates ferroptosis—a non-apoptotic, iron-dependent form of regulated cell death intrinsically linked to toxic oxidative stress (5). In the central nervous system, heightened oxidative stress and ferroptotic pathways are critical factors in the pathogenesis of neurodegenerative and psychiatric disorders (6). Significant alterations in antioxidant enzyme activities, such as myeloperoxidase (MPO), malondialdehyde (MDA), and catalase (CAT), have been identified as highly reliable diagnostic biomarkers in conditions such as schizophrenia and generalized anxiety disorder (6). By mapping these granular, sub-cellular mechanisms, the medical and scientific communities can identify the specific pathways through which oxidative stress initiates and sustains tissue degradation, muscular sarcopenia, metabolic dysfunction, and cellular senescence (3). Recent studies emphasize that recognizing the complex interplay between genetic determinants and these oxidative stress triggers is essential for developing personalised redox-modulating interventions(7).
3.Clinical Summary
Clinicians must shift their paradigm to recognize that oxidative stress is not merely a secondary byproduct or symptom of chronic disease, but rather a foundational, initiating driver of conditions ranging from the hematological (e.g., CKD-induced anemia via red blood cell hemolysis) to the psychiatric (e.g., schizophrenia characterized by demonstrably elevated MDA and MPO biomarkers) (4). Furthermore, the relatively recent identification of iron-dependent ferroptosis opens novel therapeutic avenues for preventing non-apoptotic cell death in neurodegenerative, cardiovascular, and metabolic diseases (5).
Preventative and therapeutic interventions require a highly disciplined, multi-modal approach. Dietary optimization utilizing antioxidant-rich foods, the aggressive reduction of environmental toxin exposure (including smoking cessation and pollution mitigation), and the strategic utilization of targeted, high-bioavailability antioxidant therapies are paramount in disrupting the reciprocal, destructive loop of oxidative stress and systemic inflammation(1). Addressing oxidative stress effectively in a clinical setting requires the careful monitoring of specific blood and tissue biomarkers—such as serum levels of superoxide dismutase, catalase, myeloperoxidase, and overall glutathione status—to evaluate the true efficacy of prescribed antioxidant interventions and to quantitatively delay the progression of age-related systemic decline and metabolic dysfunction.6
4.Lay Summary
To understand oxidative stress, it is helpful to think of a process similar to rust forming on a piece of metal, but happening invisibly inside your body's cells.
Just as oxygen and moisture slowly degrade a car's exterior over years of exposure, unstable, highly reactive molecules in your body—commonly called "free radicals"—damage your healthy cells over time. These free radicals are produced naturally every second as your cells burn fuel to generate energy, but their numbers can skyrocket to dangerous levels due to outside factors like air pollution, poor diet, smoking, chemical exposure, and UV radiation.
Normally, your body is equipped with a built-in defense system of "antioxidants" that intercept and neutralize these free radicals before they can do any permanent harm. However, as we age, or when we are exposed to too many environmental toxins, the sheer volume of free radicals overwhelms the body's natural defense system. When this happens, the free radicals act like microscopic vandals, attacking vital cellular machinery, breaking down healthy fats, disabling proteins, and even causing mutations in your DNA. This microscopic "rusting" process triggers widespread inflammation as the immune system attempts to clean up the damage. Unfortunately, this inflammation creates even more free radicals, trapping the body in a destructive, endless cycle. This cycle accelerates the physical signs of aging and serves as the starting point for a vast array of serious illnesses, including heart disease, atherosclerosis, kidney failure, muscle loss, and even mental health conditions like anxiety and schizophrenia. Stopping or slowing down this rusting process requires limiting your exposure to environmental toxins and actively supporting the body's defense systems with proper nutrition and specific medical interventions.
5.Essential findings
Categorization of Triggers: The induction of oxidative stress is categorized into internal and external domains. Internal mechanisms include mitochondrial electron transport chain dysfunction and hyperactive NADPH oxidase systems. External precipitants encompass environmental pollution, UV radiation, chemical exposure, smoking, and poor nutritional intake / including excessive consumption of some food groups at the expenses of others (2).
The Chronic Inflammation Loop: Oxidative cellular damage acts as an immunological trigger. The subsequent inflammatory response utilizes ROS as a weapon against perceived threats, creating a highly destructive, self-perpetuating cycle where chronic inflammation and reactive oxygen species generation continuously amplify one another, accelerating overall tissue damage and aging (4).
Erythrocyte Degradation and Anemia: Oxidative stress directly oxidizes the delicate lipid bilayers of red blood cells. This oxidation diminishes membrane elasticity and promotes premature hemolysis, acting as a primary, non-erythropoietin-dependent driver of anemia, particularly within aging populations and those suffering from advancing Chronic Kidney Disease (CKD)(4).
Ferroptotic Cell Death Pathways: The dysregulation of iron homeostasis leads to an expanded labile iron pool (LIP). Unbound iron propagates lethal oxidative stress via Fenton reactions, initiating ferroptosis—a programmed cell death pathway distinct from apoptosis. This pathway is frequently triggered by molecules acting on voltage-dependent anion channels (VDACs) and is deeply implicated in neurological and metabolic decline (5).
Psychiatric Diagnostic Biomarkers: Oxidative stress markers serve as potent, quantifiable diagnostic tools in modern psychiatry. Clinical studies reveal significantly elevated levels of malondialdehyde (MDA) and myeloperoxidase (MPO), alongside altered catalase (CAT) activity, in patients diagnosed with schizophrenia. Combined receiver operating characteristic (ROC) curves utilizing these specific biomarkers yield an area under the curve (AUC) of 0.995, indicating extraordinarily high diagnostic precision and highlighting the biological basis of psychiatric disorders (6).
Metabolic and Hepatic Induction: In the context of Non-Alcoholic Fatty Liver Disease (NAFLD), systemic perturbations in lipid metabolism force intracellular organelles—specifically mitochondria and the endoplasmic reticulum—to generate acid radicals and excessive ROS. This internal oxidative storm drives the disease's aggressive progression from benign fat accumulation to Non-Alcoholic Steatohepatitis (NASH) (8).
Pathology of Premature Aging Syndromes: In Werner syndrome, a rare genetic premature aging disease, severe, unmitigated oxidative stress and subsequent cellular dysfunction lead to distinct, visible pathological features. These include the abnormal accumulation of calcium phosphate and extreme lymphatic vessel remodeling within subcutaneous tissues, directly resulting in painful, refractory skin ulcers (9).
Gynecological and Reproductive Pathologies: In endometriosis, a painful condition characterized by ectopic endometrial tissue implantation, chronic inflammation is sustained by a profound redox imbalance. This oxidative stress is evidenced by significantly decreased systemic levels of essential antioxidant trace elements, such as zinc (Zn), highlighting the role of oxidative damage in reproductive diseases and infertility (10).
Skeletal Muscle Degradation: Reactive oxygen and nitrogen species fundamentally alter skeletal muscle physiology. While low levels are required for exercise adaptation, chronic ROS overexposure drives the pathogenesis of sarcopenia, cachexia, and metabolic myopathies through the destructive modification of redox-sensitive signaling pathways (3).
References
Liguori, I. et al. "Oxidative stress, aging, and diseases." Clinical Interventions in Aging, 2018.
Pizzino, G. et al. "Oxidative Stress: Harms and Benefits for Human Health." Oxidative Medicine and Cellular Longevity, 2017.
Cabello-Verrugio, C. et al. "Oxidative Stress in Disease and Aging Mechanisms." Antioxidants, 2020.
Ighodaro, O.M. et al. "First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX)." Alexandria Journal of Medicine, 2018.
Sureshbabu, A. et al. "Oxidative stress, age-related diseases, and relative biomarkers." Aging, 2015.
Dixon, S.J. et al. "Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death." Cell, 2012.
Smaga, I. et al. "Oxidative stress and aging psychiatric diagnostic biomarkers." Brain Sciences, 2021.
Gao, Q. et al. "Oxidative stress: from molecular studies to clinical intervention strategies." Frontiers in Molecular Biosciences, 2025.
Chen, Z. et al. "Oxidative stress and aging NAFLD NASH essential findings." PMC, 2020.
Takemoto, M. et al. "Oxidative stress and aging Werner Syndrome essential findings." Aging-US, 2024.
Bugajska, J. et al. "Pathophysiology and treatment of Endometriosis Systematic Review." Antioxidants, 2024.